Cancer Immunoactivation & Suprresion of Neuroinflammation
1. Enhancement of Splice-Neoantigen Expression and Cancer Immunoactivation by RECTAS
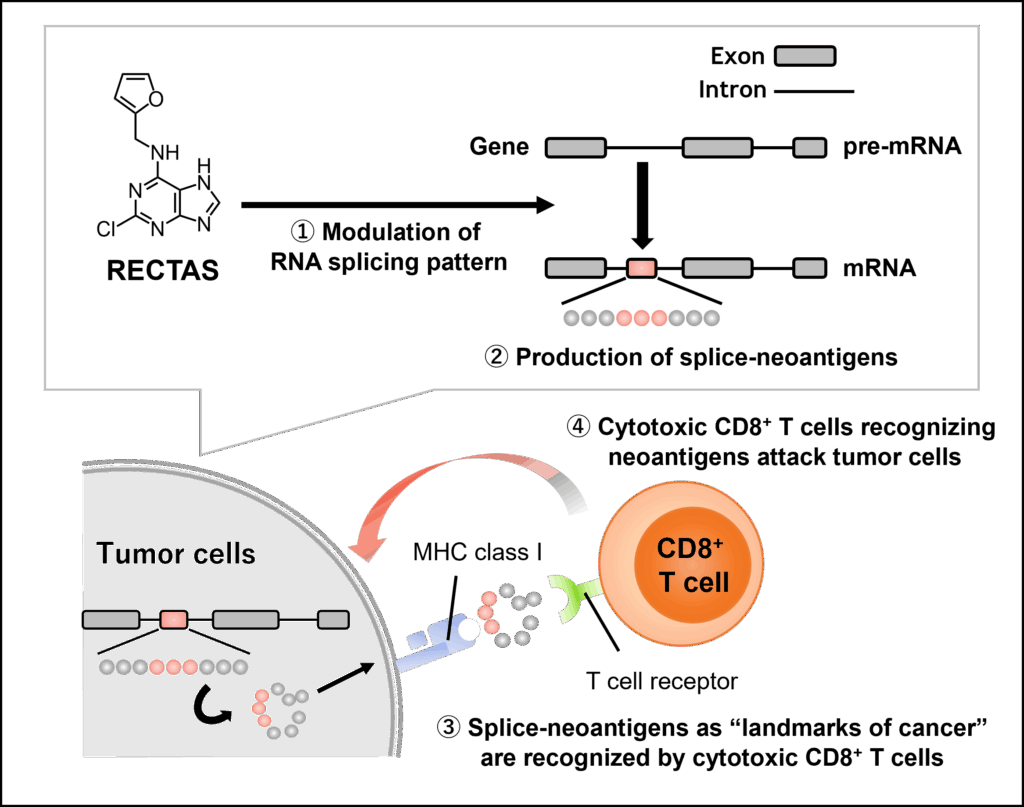
The generation of neoantigens is a key determinant of the efficacy of cancer immunotherapy. However, increasing the abundance of neoantigens in order to achieve more effective cancer treatment remains technically challenging. We found that RECTAS, a splicing-modulating compound independently developed by our group, can induce the production of splice neoantigens that may contribute to the enhancement of antitumor immune responses (Matsushima et al., Sci Transl Med. 2022). RECTAS suppressed tumor growth in a CD8+ cell– and tumor major histocompatibility complex class I–dependent manner and enhanced the efficacy of immune checkpoint blockade. Subsequent transcriptome analyses and immunogenicity validation experiments identified six candidate splice neoantigens whose expression was induced by RECTAS treatment. Vaccination with the identified neoepitopes elicited T-cell responses capable of killing cancer cells in vitro, and sensitization with RECTAS led to further suppression of tumor growth in vivo. Together, these findings support the potential of developing therapeutic strategies that enhance the efficacy of cancer immunotherapy through the induction of splice variants.
2. Discovery of ALGERNON2, a Novel Therapeutic Candidate for the Suppression of Neuroinflammation
The brain contains not only neurons, which are responsible for higher-order functions, but also glial cells, which support neuronal function. Among these, microglia serve as the immune system of brain tissue: they remove pathogens and unwanted materials through phagocytosis and enhance immune responses by releasing cytokines in response to injury. Under pathological conditions, such as brain injury and neurodegenerative disease, excessive activation of microglia leads to increased cytokine production and results in a state of neuroinflammation. Neuroinflammation is recognized as one of the hallmarks of progressive neurodegenerative diseases, including Alzheimer’s disease and Parkinson’s disease, and has also been observed in depression and depressive symptoms, suggesting its involvement in these conditions. In progressive neurodegenerative diseases in particular, neurons are gradually lost over time, yet no breakthrough therapy has been established, despite extensive development efforts by major pharmaceutical companies worldwide.
In the course of screening for compounds that promote the proliferation of neural stem cells, which is reduced in Down syndrome, we identified ALGERNON (altered generation of neurons) (Kobayashi et al., Proc Natl Acad Sci U S A. 2017). ALGERNON promoted the proliferation of neural stem cells derived from human Down syndrome iPS cells and, when administered to pregnant mice, improved cognitive function in offspring with Down syndrome. We subsequently succeeded in developing ALGERNON2, a compound with improved safety, more favorable pharmacokinetic properties, and enhanced penetration into brain tissue (Kobayashi et al., Sci Adv. 2020). When we examined the pharmacological effects of ALGERNON2 in adult mice, in comparison with the developmental stage of the brain when neural stem cell proliferation is highly active, we unexpectedly found that ALGERNON2 suppresses cytokine production by microglia and thereby inhibits neuroinflammation (Kobayashi et al., Proc Natl Acad Sci U S A. 2023). ALGERNON2 has also been shown to improve the engraftment of transplanted iPS cell–derived dopaminergic neural precursor cells by suppressing inflammation in the brain (Patent Publication No. WO/2021/201171).
