Transcriptome-Based Drug Discovery
At the Hagiwara Laboratory, Department of Drug Discovery Medicine, we promote transcriptome-based drug discovery, in which therapeutic efficacy is evaluated and mechanisms of action are identified using transcriptome analysis and gene expression regulation as key indicators. Through this approach, we have advanced the development of small-molecule compounds that we have discovered—including hit compounds and lead compounds—to the stage of investigator-initiated clinical trials. The following projects are underway as part of our efforts to address major societal challenges facing humanity.
1. FIT039, a Candidate Anti-HPV Therapeutic for the Treatment of Warts and the Prevention of Cervical Cancer
FIT039, a CDK9 inhibitor being co-developed with KinoPharma Inc., an academia-originated venture company, suppresses the transcription of a broad range of DNA viruses, including papillomaviruses, herpesviruses, adenoviruses, and hepatitis B virus, as well as human immunodeficiency virus (HIV), thereby inhibiting their replication (Yamamoto et al., J Clin Invest. 2014; Ajiro et al., Clin Cancer Res. 2018). In clinical trials, FIT039 patches and ointments demonstrated a wart-reducing effect. In addition, FIT039 vaginal tablets showed in a Phase II clinical trial that human papillomavirus (HPV) could be eliminated from the cervix. FIT039 is therefore expected to make a substantial contribution to public health as a new therapeutic that could help prevent cervical cancer in individuals who have not received HPV vaccination.
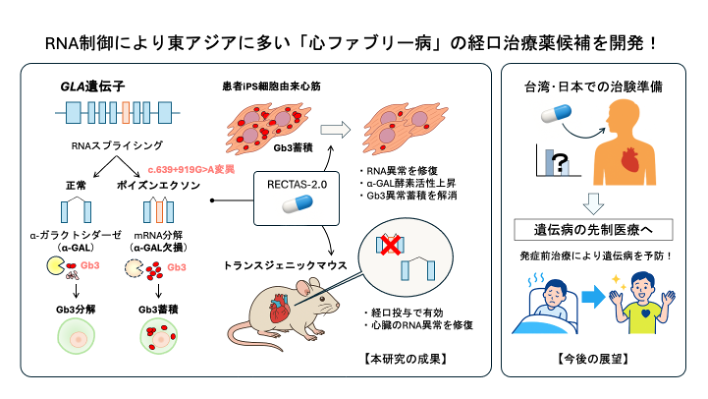
2. RECTAS2.0, a Candidate Preemptive Therapeutic for Inherited Disorders
Cardiac Fabry disease is an inherited disorder caused by the GLA c.639+919G>A mutation, which is particularly prevalent in East Asia, including Japan, and leads to cardiac dysfunction after middle age. Using cardiomyocytes generated from patient-derived iPS cells, we developed RECTAS2.0, a compound that corrects aberrant RNA splicing, and successfully restored enzyme activity (Awaya et al., Sci Adv. 2025). Oral administration of this compound also corrected the splicing abnormality in the myocardium of genetically engineered mice. Unlike conventional enzyme replacement therapy, this compound is orally available and readily reaches cardiac tissue. It is therefore expected to enable the realization of preemptive RNA medicine, in which treatment can be initiated at an early stage in carriers identified by genetic diagnosis, thereby preventing the onset of inherited disease (see the figure below and the press release). Since it is estimated that more than 10,000 patients with cardiac Fabry disease who may be treatable with this compound reside in western Japan and Taiwan, we are working toward practical application in collaboration with BTB Therapeutics, Inc., a Kyoto University venture company, and MagMIT Pharmaceutical Co., Ltd. In addition to cardiac Fabry disease, RECTAS2.0 is also expected to exert similar therapeutic effects in other inherited disorders caused by aberrant RNA splicing.
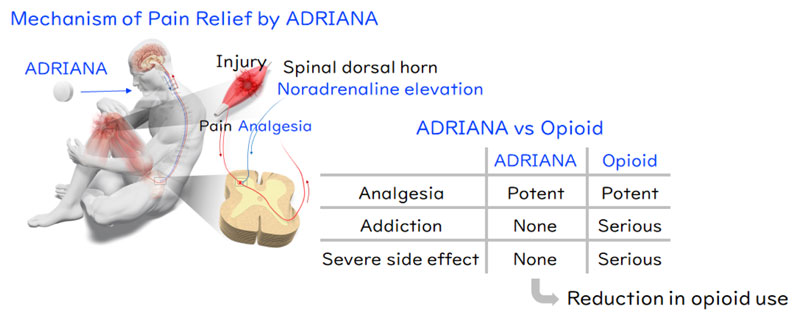
3. ADRIANA, a Candidate Non-Opioid Analgesic Without Serious Adverse Effects or Dependence
In the United States, overdose of synthetic narcotics (opioids) such as fentanyl claimed as many as 80,000 lives in 2023, in what has come to be known as the opioid crisis. For this reason, the Trump administration has pressed neighboring countries to strengthen border security; however, a fundamental solution will require the development of new therapeutics that can replace opioid analgesics. Inspired by the fact that animals, including humans, suppress pain by releasing noradrenaline from neurons when faced with life-threatening situations, we identified ADRIANA, a groundbreaking analgesic that acts through a mechanism entirely distinct from that of opioids (Toyomoto et al., Proc Natl Acad Sci U S A. 2025). ADRIANA exhibits analgesic efficacy comparable to that of morphine against severe pain, including cancer pain, while showing neither dependence nor serious adverse effects, and therefore has the potential to become an innovative therapeutic capable of helping society overcome the opioid crisis. Highly promising results have been obtained in an investigator-initiated clinical trial of ADRIANA conducted at Kyoto University Hospital, and preparations are underway, in collaboration with BTB Therapeutics, Inc., for a large-scale Phase II clinical trial in the United States (see the figure below and the press release).